
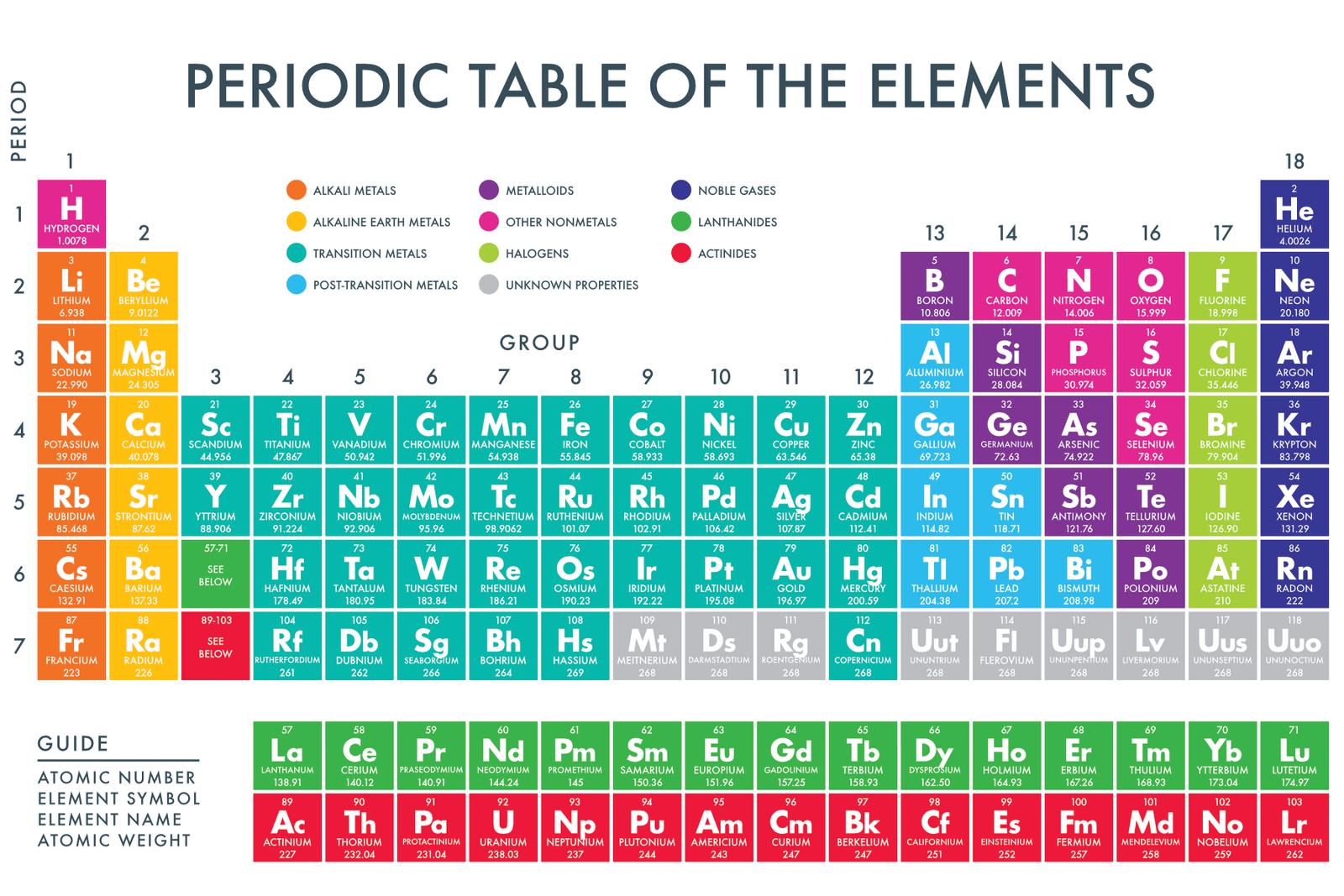
The same goes for the isolated lower portion of the Periodic Table (also "blacked out" in the figure of the Periodic Table above). They behave differently, and you can't apply the same rules to them as far as valence electrons are concerned. The elements in that lower portion of the Periodic Table (the middle portion of the Periodic Table "blacked out" in the first figure) are known as transition metals. This rule doesn't apply, however, to the Group 1B–8B (also known as Groups 3–12) elements. Our rule for determining the energy level of an element's valence electrons using the element's row in the Periodic Table works for Group 1A–8A elements. You should make note of one final point when it comes to energy levels and how they relate to the Periodic Table. Similarly, Iodine (I) is found in row 5 of the Periodic Table, and its valence electrons are found in the n = 5 energy level. Chlorine (Cl), for instance, is found in row 3 of the Periodic Table, and its valence electrons are found in the n = 3 energy level. Once again, an element's row can be used to determine the energy level of that element's valence electrons. If we write the electron configuration for the Group 1A metal from each row of the Periodic Table, we have:ġ s 2 2 s 2 2 p 6 3 s 1 To understand what this means in terms of an element's electron configuration, let's consider the Group 1A metals. The transition metals and the lanthanides and actinides have been omitted. The figure below shows how the different rows in the Periodic Table are numbered. Rows Across on the PT are Consistent With the Energy Level in An Atom įirst, let's try to figure out what we can learn from an element's row or period in the Periodic Table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
